Cathetered-Directed Therapy for "Intermediate-Risk PE"
A "positive" trial hiding the truth behind a composite primary outcome.
Treatment of acute pulmonary embolism spans a continuum between “watchful waiting” and heroic interventions. In the middle, you have non-crashing patients with tenuous clinical stability, and options like catheter-directed thrombolysis.
In this vein HI-PEITHO is a trial deploying CDL for those “intermediate risk" PEs, as compared to a traditional strategy of systemic anticoagulation and observation. The primary outcome is a very impressive sounding composite of:
PE-related mortality, cardiorespiratory decompensation or collapse, or nonfatal symptomatic and objectively confirmed recurrence of PE, within seven days of randomization.
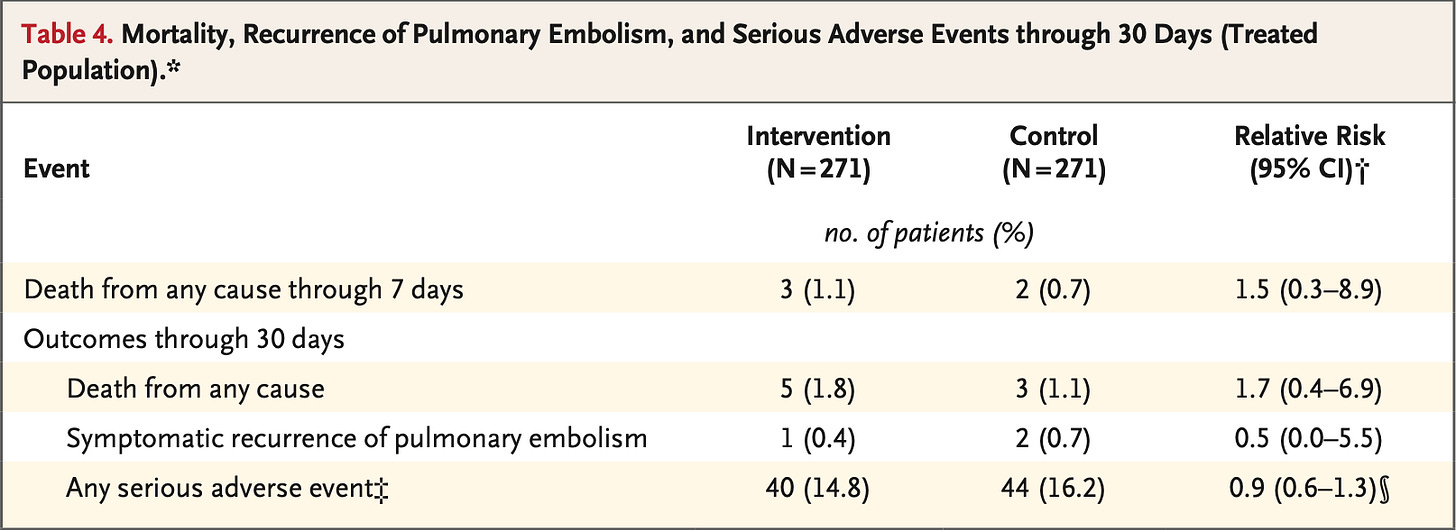
And, good news for the sponsors at Boston Scientific, the patients randomized to their fancy CDL did not suffer as many primary outcomes, and – emphasised sweetly in the text and the editorial – this was driven by reductions in “cardiorespiratory decompensation”. Cardiorespiratory decompensation sounds like a good thing to avoid!
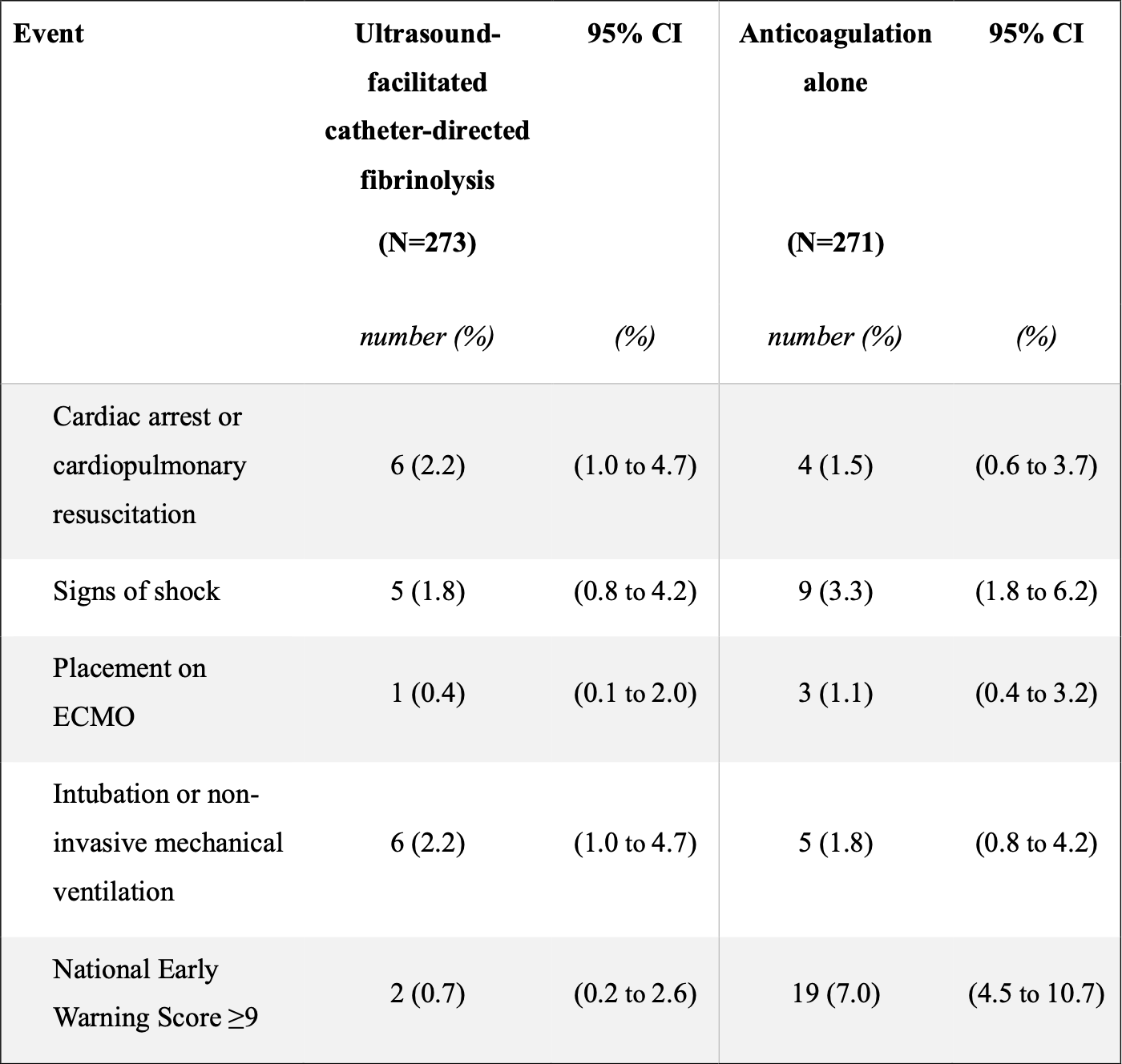
This is what “cardiorespiratory decompensation” looked like in the two groups:
So, mostly small numbers and grossly “the same”, considering – except for the NEWS score. At the end of the day, the only Real Difference in the primary outcome was a few more patients with increased physiologic stress within 24 hours of randomisation.
What happened to these folks? Well, they went off and received a variety of rescue therapies – mostly a type of CDL, embolectomy, or systemic thrombolysis. And they did fine, regardless of whether they had the blanket early intervention, or watchful waiting with selective intervention:
So, what this trial is really telling us: there's no reason to do the special CDL on everyone because you can watch out for the ~5-10% of patients who get worse, and then provide them with an appropriate escalation in therapy.
Unfortunately, that doesn’t allow Boston Scientific to sell more catheters!